Understanding the Electron Configuration Ge is primal to savvy the doings and properties of germanium, a crucial element in the semiconductor industry. Germanium, with the atomic turn 32, is a metalloid that plays a significant role in diverse technical applications, particularly in electronics and optics. This blog post delves into the Electron Configuration Ge, its significance, and how it influences the element's chemical and physical properties.
What is Electron Configuration?
Electron configuration refers to the arrangement of electrons in the orbitals of an atom. It is a critical concept in chemistry that helps explicate the periodic trends and chemic behaviour of elements. The configuration is typically pen using a notation that specifies the energy levels (n) and the type of orbital (s, p, d, f) along with the figure of electrons in each orbital.
Electron Configuration of Germanium
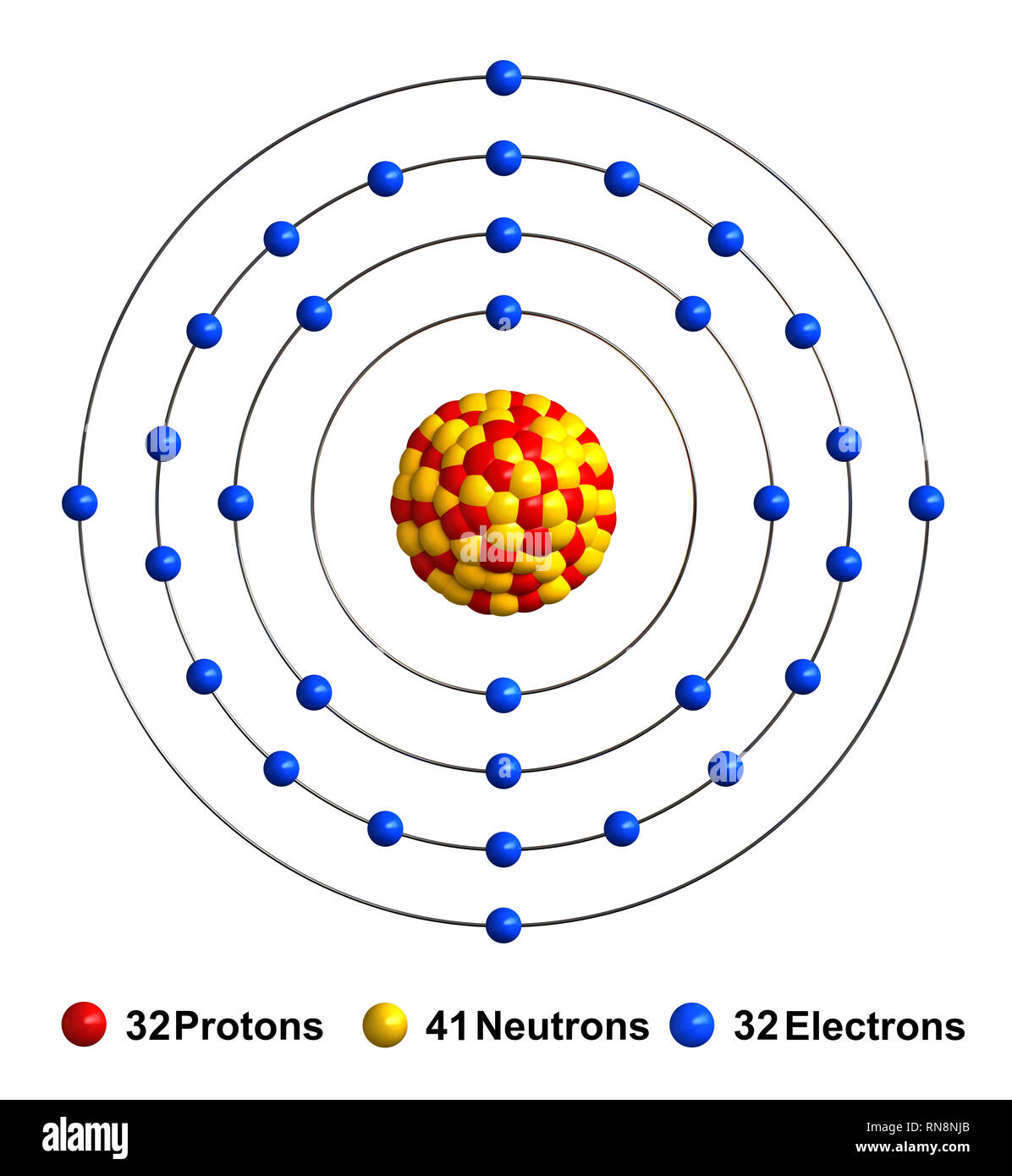
The Electron Configuration Ge can be shape by following the Aufbau principle, which states that electrons fill the lowest energy orbitals first. Germanium has 32 electrons, and its electron configuration is:
1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 2
Breaking this down:
- 1s 2: The first energy point contains 2 electrons in the s orbital.
- 2s 2 2p 6: The second energy tier contains 2 electrons in the s orbital and 6 electrons in the p orbital.
- 3s 2 3p 6: The third energy level contains 2 electrons in the s orbital and 6 electrons in the p orbital.
- 4s 2 3d 10 4p 2: The fourth energy level contains 2 electrons in the s orbital, 10 electrons in the d orbital, and 2 electrons in the p orbital.
This configuration shows that germanium has a partially filled 4p orbital, which is substantial for its chemical reactivity and electronic properties.
Significance of Electron Configuration in Germanium
The Electron Configuration Ge has respective implications for the element s properties and applications:
- Semiconducting Properties: Germanium s part filled 4p orbital allows it to act as a semiconductor. Semiconductors have electrical conductivity between that of a director and an dielectric, get them ideal for electronic devices.
- Chemical Reactivity: The electron shape influences germanium s reactivity. It can form compounds with several elements, including oxygen, sulfur, and halogens, which are useful in different industrial applications.
- Optical Properties: Germanium s electronic construction also affects its optical properties. It is transparent to infrared light, making it utilitarian in infrared optics and night vision devices.
Applications of Germanium
Germanium s unique properties, staunch from its Electron Configuration Ge, get it valuable in several industries:
- Electronics: Germanium was one of the first materials used in transistors and diodes. Although silicon has largely supersede it, germanium is still used in eminent frequency and eminent power applications.
- Optics: Germanium s transparency to infrared light makes it ideal for lenses and windows in infrared cameras and other optical devices.
- Fiber Optics: Germanium is used as a dopant in fiber optics to heighten the refractive index of the fibre, improving its performance.
- Solar Cells: Germanium is used in multi junction solar cells, which are highly effective and used in space applications.
Comparing Germanium with Other Semiconductors
To better understand the signification of germanium s Electron Configuration Ge, it is helpful to compare it with other semiconductors, such as silicon and gallium arsenide.
| Element | Electron Configuration | Key Properties |
|---|---|---|
| Germanium (Ge) | 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 2 | Semiconducting, transparent to infrared light |
| Silicon (Si) | 1s 2 2s 2 2p 6 3s 2 3p 2 | Semiconducting, widely used in electronics |
| Gallium Arsenide (GaAs) | Ga: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 1 As: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3 |
Semiconducting, used in high speed electronics and optoelectronics |
Each of these elements has a unequaled electron configuration that contributes to its specific properties and applications. Germanium s part occupy 4p orbital gives it distinct advantages in certain areas, such as infrared optics and eminent frequency electronics.
Note: The electron configuration of gallium arsenide is shown for both gallium and arsenic severally, as it is a compound semiconductor.
Future Prospects of Germanium
Despite the dominance of silicon in the semiconductor industry, germanium continues to be a material of interest for future technologies. Its unparalleled properties make it a candidate for advanced applications in electronics, optics, and photonics. Research is ongoing to explore new ways to utilise germanium s Electron Configuration Ge to germinate more effective and powerful devices.
One area of particular interest is the development of germanium found transistors for eminent speed and low ability applications. Germanium's higher electron mobility compared to silicon makes it a promising material for next coevals transistors. Additionally, germanium's compatibility with silicon technology allows for the consolidation of germanium devices with live silicon based circuits, paving the way for hybrid electronic systems.
In the field of optics, germanium's transparency to infrared light makes it a worthful material for acquire advanced opthalmic components. Researchers are exploring the use of germanium in infrared detectors, lasers, and other optical devices for applications in telecommunications, medical imaging, and environmental monitoring.
Furthermore, germanium's potential in photovoltaics is being enquire. Germanium based solar cells have shown high efficiency and are being studied for use in multi colligation solar cells, which are subject of converting a broader spectrum of sunlight into electricity. This makes germanium an important material for hereafter solar energy technologies.
to summarize, the Electron Configuration Ge plays a crucial role in set the properties and applications of germanium. Its singular electronic structure makes it a valuable material in respective industries, from electronics and optics to photovoltaics. As research continues to uncover new applications and improve survive technologies, germanium s significance in mod skill and orchestrate is probable to turn. Understanding the Electron Configuration Ge is essential for harnessing the total likely of this remarkable element and developing advanced solutions for the challenges of the futurity.
Related Terms:
- germanium electron configuration entire
- ge full electron constellation
- germanium ground state electron form
- germanium electron shape chart
- electron configuration for germanium
- electron conformation for germanium ge