Chemistry is a intrigue battleground that delves into the intricacies of subject and its interactions. One of the rudimentary concepts in chemistry is the study of chemic bonds and the energy changes that occur during their formation and separate. Understanding whether breaking bonds is heat-absorbing or exothermic is crucial for savvy the energetics of chemic reactions. This post will explore the concept of bond breaking, the energy involve, and the signification of know whether break bonds is endothermic.
Understanding Chemical Bonds
Chemical bonds are the forces that hold atoms together in molecules or crystals. There are several types of chemical bonds, including ionic, covalent, and metallic bonds. Each type of bond has its unequaled characteristics and energy requirements. For representative, covalent bonds involve the sharing of electrons between atoms, while ionic bonds imply the transferee of electrons from one atom to another.
Energy Changes in Chemical Reactions
Chemical reactions involve the separate and organize of chemic bonds. These processes are accompany by energy changes. The energy required to break a bond is known as bond dissociation energy. Conversely, the energy release when a bond is spring is known as bond formation energy. The overall energy change in a reaction is the difference between the energy involve to break bonds and the energy released when new bonds are formed.
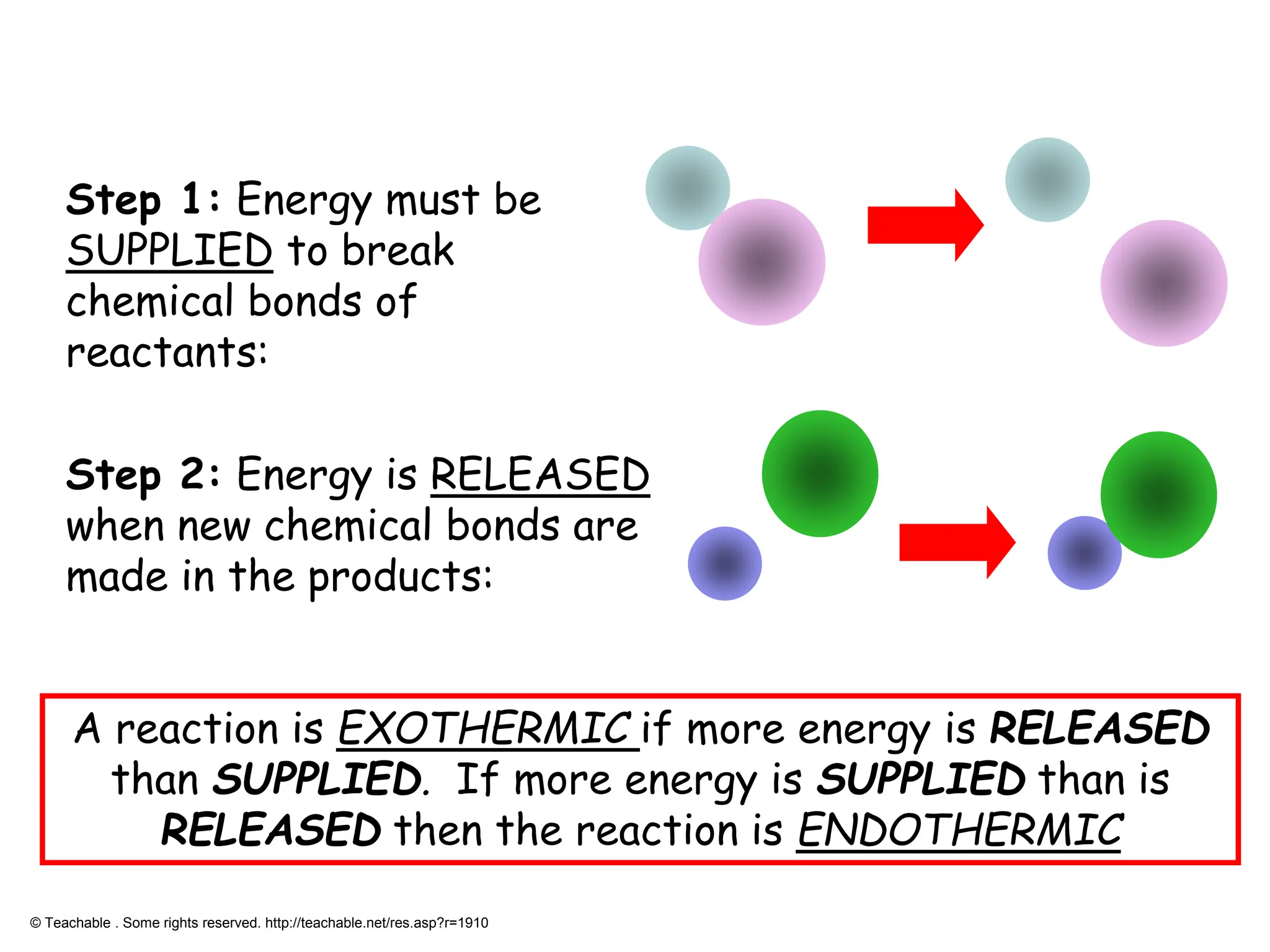
Is Breaking Bonds Endothermic?
To determine whether breaking bonds is heat-absorbing, we need to interpret the definition of endothermal processes. An endothermic process is one that absorbs heat from its surroundings. When bonds are break, energy is take to overcome the attractive forces between atoms. This energy is typically supplied in the form of heat, make the operation endothermic.
for instance, consider the disassociation of a water molecule (H 2 O) into hydrogen (H2 ) and oxygen (O2 ) gases. The reaction can be represented as:
H 2 O (l) → H2 (g) ½ O 2 (g)
This reaction requires energy to break the covalent bonds between hydrogen and oxygen atoms. The energy assimilate during this process makes it endothermic.
Energy Requirements for Bond Breaking
The energy involve to break a bond depends on the type of bond and the specific atoms involved. For illustration, covalent bonds generally command more energy to break than ionic bonds. The bond disassociation energy is a quantify of the strength of a chemical bond and is typically verbalize in kilojoules per mole (kJ mol).
Here is a table showing the bond dissociation energies for some common bonds:
| Bond Type | Bond Dissociation Energy (kJ mol) |
|---|---|
| H H | 436 |
| O O | 498 |
| C H | 413 |
| C C | 614 |
| C O | 360 |
These values indicate the amount of energy needed to break one mole of the specified bonds. Understanding these energies is essential for predicting the energetics of chemic reactions.
Factors Affecting Bond Breaking
Several factors influence the energy demand to break chemical bonds. These include:
- Bond Strength: Stronger bonds take more energy to break. for instance, triple bonds are stronger than double bonds, which in turn are stronger than single bonds.
- Atomic Size: Larger atoms mostly form weaker bonds because their valence electrons are farther from the nucleus, prima to less efficacious overlap.
- Electronegativity: The difference in negativity between atoms affects bond strength. Bonds between atoms with similar electronegativities are broadly stronger.
- Environmental Conditions: Factors such as temperature and pressing can also influence the energy required to break bonds. Higher temperatures can provide the necessary energy to break bonds more easy.
Applications of Bond Breaking
The concept of bond breaking and the energy involved has numerous applications in various fields. For illustration, in industrial chemistry, read bond breaking is essential for plan effective chemical processes. In biochemistry, the interrupt and form of bonds are essential for biological processes such as metamorphosis and enzyme catalysis.
In environmental skill, the separate of chemic bonds is important for understand the abasement of pollutants and the motorcycle of nutrients. for instance, the breakdown of organic compounds in soil and water involves the break of covalent bonds, which is oft facilitate by microorganisms.
In materials skill, the strength of chemic bonds determines the properties of materials. For example, the high bond disassociation energy of diamond makes it one of the hardest materials known. Understanding bond separate is all-important for developing new materials with desired properties.
Note: The energy involve to break bonds is a critical element in determining the feasibility of chemic reactions. Reactions that expect a lot of energy to break bonds may not be ad-lib and may ask outside energy input.
Conclusion
In compact, interrupt chemical bonds is an endothermic process that requires energy to overcome the attractive forces between atoms. The energy demand to break bonds, known as bond dissociation energy, varies depending on the type of bond and the specific atoms involve. Understanding whether interrupt bonds is endothermal is crucial for predicting the energetics of chemical reactions and has legion applications in several fields, including industrial chemistry, biochemistry, environmental science, and materials skill. By grasping the fundamentals of bond breaking and the energy imply, we can gain deeper insights into the complex domain of chemic interactions.
Related Terms:
- bond energy calculations
- do exothermic reactions make bonds
- interrupt chemic bonds
- is bond get endothermal
- heat-absorbing chemical reactions
- breaking do chemistry