In the realm of physics and direct, the concept of Units of J is fundamental. Units of J refer to the joule, the standard unit of energy in the International System of Units (SI). Named after the English physicist James Prescott Joule, the joule is a crucial measurement in diverse scientific and engineering disciplines. Understanding Units of J is essential for anyone working in fields that regard energy, work, or heat.
Understanding the Joule
The joule is delimit as the amount of work done when a force of one newton acts over a distance of one measure. In numerical terms, one joule is equal to one newton meter (N m). This definition highlights the relationship between force, length, and energy, which is a cornerstone of classic mechanics.
To put it simply, if you apply a force of one newton to travel an object one metre in the way of the force, you have done one joule of work. This concept is wide used in various applications, from reckon the energy ask to lift an object to determining the energy output of a machine.
Applications of Units of J
The joule is used in a wide range of applications across different fields. Some of the most common applications include:
- Mechanical Work: In mechanical engineering, the joule is used to mensurate the act done by a machine or the energy transplant from one object to another.
- Electrical Energy: In electrical mastermind, the joule is used to measure the energy transferred by an electric current. for instance, one joule is the energy dissipated when a current of one ampere passes through a resistivity of one ohm for one second.
- Thermodynamics: In thermodynamics, the joule is used to quantify heat energy. The specific heat capacity of a meat, for example, is much expressed in joules per kilogram per degree Celsius (J kg C).
- Chemistry: In chemistry, the joule is used to quantify the energy liberate or ingest in chemic reactions. The enthalpy vary of a reaction, for instance, is often verbalise in kilojoules per mole (kJ mol).
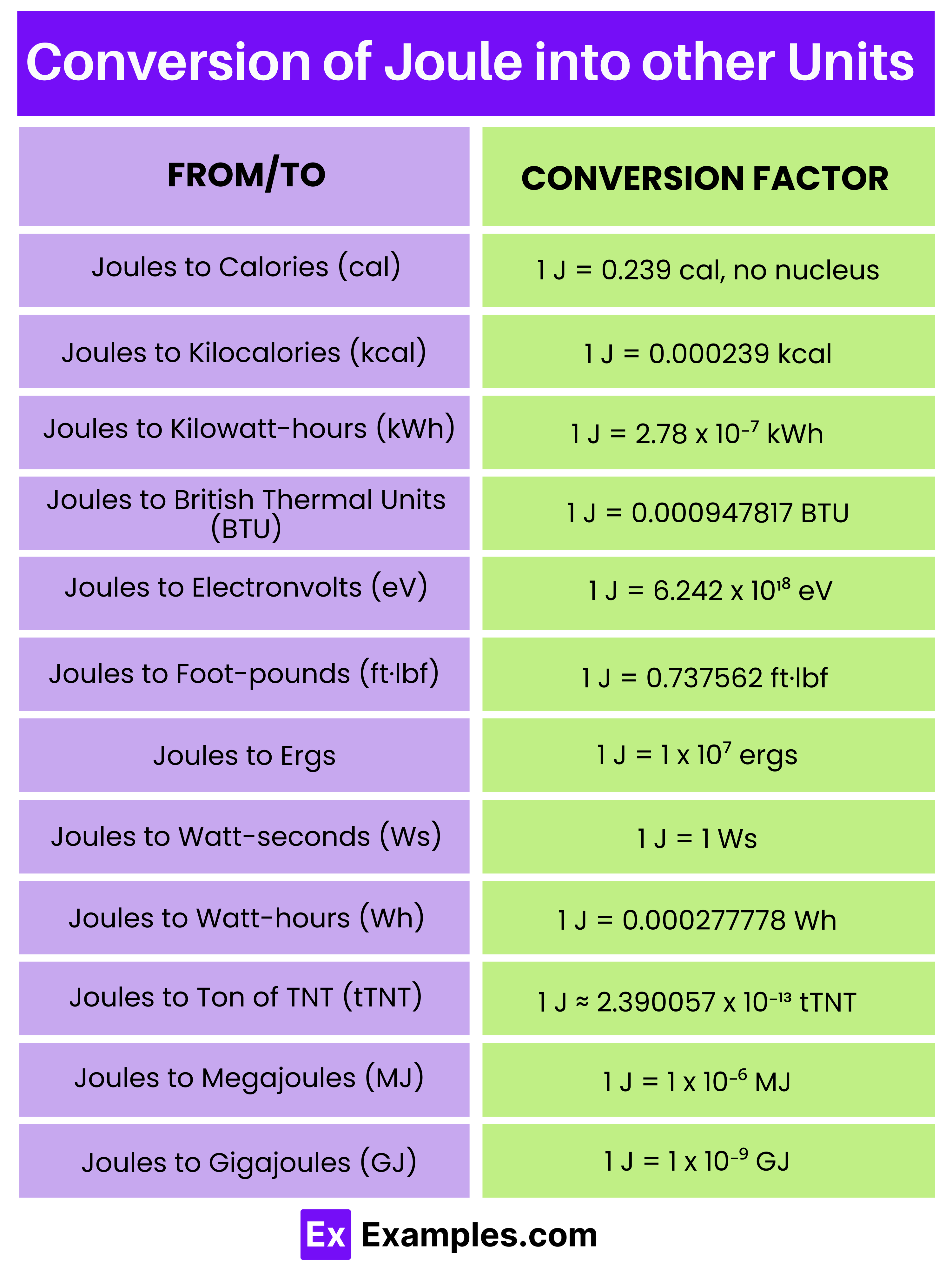
Conversions Involving Units of J
Understanding how to convert between different units of energy is all-important for many applications. Here are some common conversions involving Units of J:
| Unit | Conversion to Joules |
|---|---|
| Calorie (cal) | 1 cal 4. 184 J |
| Kilocalorie (kcal) | 1 kcal 4184 J |
| Electronvolt (eV) | 1 eV 1. 602 10 19 J |
| British Thermal Unit (BTU) | 1 BTU 1055. 06 J |
| Watt hour (Wh) | 1 Wh 3600 J |
These conversions are essential for equate energy values across different systems and disciplines. for illustration, in aliment, energy is often measured in kilocalories, while in electronics, energy is measured in electronvolts.
Note: When convert between units, it is significant to secure that the conversion factors are accurate and earmark for the context. Incorrect conversions can lead to important errors in calculations.
Energy and Power
While the joule measures energy, ability is the rate at which energy is transferred or converted. Power is measured in watts (W), where one watt is equal to one joule per second (J s). Understanding the relationship between energy and power is crucial for many applications, such as designing efficient machines or optimizing energy usage.
for instance, if a machine does 100 joules of work in 10 seconds, its ability output is 10 watts. This relationship is evince mathematically as:
Power (W) Energy (J) Time (s)
This equation is fundamental in various fields, from mechanical engineering to electrical organize. It allows engineers to design systems that are both efficient and effectual in their energy use.
Energy Conservation
One of the most significant principles in physics is the law of preservation of energy, which states that energy cannot be created or destroyed, only transformed from one form to another. This principle is crucial for understanding how energy flows through systems and how it can be conserved.
for instance, in a mechanical scheme, the potential energy of an object can be converted into kinetic energy as it falls. The full energy of the scheme remains constant, but the form of the energy changes. This principle is applied in several fields, from project energy effective buildings to optimizing industrial processes.
In electric systems, energy conservation is equally significant. for representative, in a circuit, the electrical energy supplied by a battery is converted into other forms of energy, such as heat or light. Understanding how to understate energy losses and maximize efficiency is crucial for designing sustainable and cost effective systems.
Note: Energy preservation is not just about trim energy consumption; it is also about optimizing the use of available energy resources. By understanding the principles of energy preservation, engineers and scientists can design systems that are both efficient and sustainable.
Energy in Everyday Life
Energy is a rudimentary part of our daily lives, and understanding Units of J helps us get sense of the world around us. From the energy we consume in food to the energy we use to power our homes and vehicles, energy is everywhere. Here are some examples of how energy is used in everyday life:
- Food Energy: The energy we get from food is mensurate in kilocalories (kcal). One kilocalorie is equal to 4184 joules. Understanding the energy substance of food helps us make inform choices about our diet and nutrition.
- Home Energy: The energy we use to ability our homes is measured in kilowatt hours (kWh). One kilowatt hour is equal to 3. 6 megajoules (MJ). Understanding how to conserve energy in our homes can help us reduce our energy bills and minimize our environmental impact.
- Transportation Energy: The energy we use to ability our vehicles is measured in assorted units, depending on the type of fuel. for instance, gasoline is often measured in gallons, while galvanising vehicles are measure in kilowatt hours. Understanding the energy efficiency of different transport methods can help us make more sustainable choices.
By understanding Units of J and how energy is used in our daily lives, we can create more inform decisions about our energy use and contribute to a more sustainable future.
Energy is a underlying part of our daily lives, and read Units of J helps us get sense of the domain around us. From the energy we consume in food to the energy we use to ability our homes and vehicles, energy is everywhere. Here are some examples of how energy is used in everyday life:

By understanding Units of J and how energy is used in our daily lives, we can make more informed decisions about our energy use and contribute to a more sustainable hereafter.
to resume, the joule is a cardinal unit of energy that plays a crucial role in various scientific and engineering disciplines. Understanding Units of J is crucial for anyone working in fields that involve energy, work, or heat. From mechanical engineering to electrical engineer, and from thermodynamics to chemistry, the joule is a versatile and significant unit of measurement. By understanding how to measure and convert energy, we can design more effective and sustainable systems, optimise our energy use, and contribute to a more sustainable future. The principles of energy conservation and the relationship between energy and ability are underlying to our understanding of the universe around us, and the joule is a key unit in this understanding.
Related Terms:
- j units breakdown
- units of j in si
- what is j in chemistry
- units of joule
- symbol for joules
- 1 j is adequate to