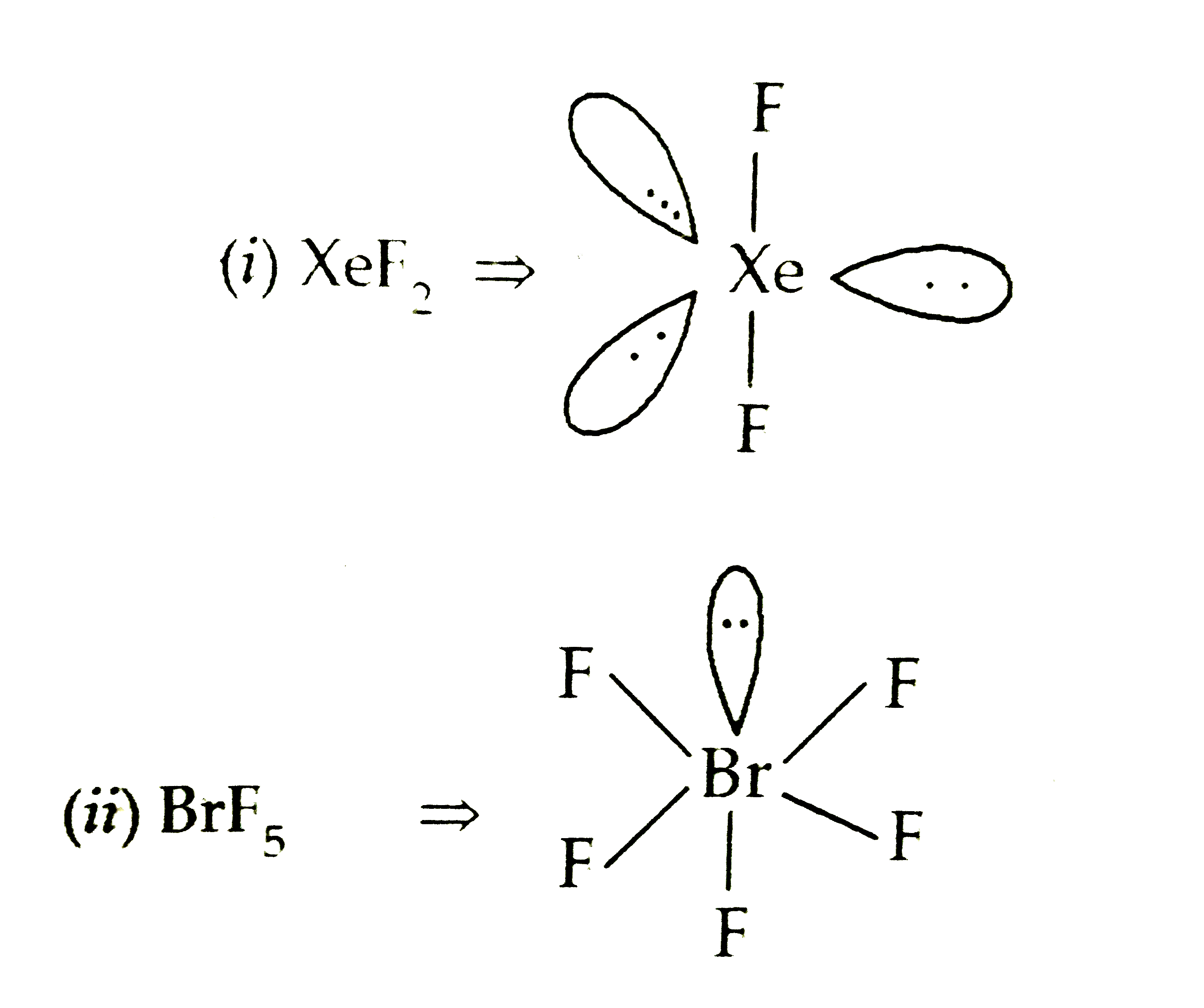Xenon difluoride (XeF2) is a beguile compound in the realm of chemistry, peculiarly known for its unparalleled properties and structure. One of the most fascinate aspects of XeF2 is its Xenon Difluoride Bond Angle, which plays a essential role in understanding its molecular geometry and reactivity. This compound is a rare model of a noble gas form a stable compound, challenging the traditional notion that noble gases are inert. In this post, we will delve into the details of XeF2, its bond angle, and the import of this angle in chemical bonding and molecular structure.
Understanding Xenon Difluoride
Xenon difluoride is a colorless, crystalline solid that is highly reactive. It was first synthesize in 1962, marking a significant milestone in the battlefield of chemistry. The compound consists of one xenon atom tie to two fluorine atoms, spring a linear molecular geometry. This linear construction is a unmediated effect of the Xenon Difluoride Bond Angle, which is 180 degrees.
The Significance of the Xenon Difluoride Bond Angle
The Xenon Difluoride Bond Angle of 180 degrees is a key factor in translate the stability and reactivity of XeF2. This bond angle indicates that the molecule is linear, with the xenon atom at the center and the two fluorine atoms on either side. The linear geometry is a event of the sp3d hybridization of the xenon atom, which allows for the shaping of two sigma bonds with the fluorine atoms.
The linear construction of XeF2 has several implications:
- The molecule is non polar, as the dipole moments of the two Xe F bonds cancel each other out.
- The linear geometry allows for efficient bundle in the solid state, contribute to its crystalline construction.
- The reactivity of XeF2 is mold by its linear structure, as the lone pairs on the xenon atom are not involved in adhere and can participate in reactions.
Molecular Orbital Theory and Xenon Difluoride
Molecular Orbital (MO) theory provides a deeper understanding of the bonding in XeF2. According to MO theory, the xenon atom contributes its 5s and 5p orbitals to form molecular orbitals with the fluorine atoms. The 5s orbital of xenon combines with the 2s orbital of fluorine to form bind and antibonding molecular orbitals. Similarly, the 5p orbitals of xenon combine with the 2p orbitals of fluorine to form bonding and antibonding molecular orbitals.
The Xenon Difluoride Bond Angle of 180 degrees is a result of the sp3d hybridizing of the xenon atom, which allows for the formation of two sigma bonds with the fluorine atoms. The linear geometry is stabilized by the overlap of the sp3d hybrid orbitals of xenon with the 2p orbitals of fluorine, organise strong sigma bonds.
Comparing Xenon Difluoride with Other Xenon Compounds
Xenon difluoride is just one of respective xenon compounds that have been synthesise. Other notable compounds include xenon tetrafluoride (XeF4) and xenon hexafluoride (XeF6). Each of these compounds has a unique molecular geometry and bond angles, which are determined by the number of fluorine atoms tie to the xenon atom and the hybridization of the xenon atom.
Here is a comparison of the bond angles in these xenon compounds:
| Compound | Molecular Geometry | Bond Angle |
|---|---|---|
| Xenon Difluoride (XeF2) | Linear | 180 degrees |
| Xenon Tetrafluoride (XeF4) | Square Planar | 90 degrees |
| Xenon Hexafluoride (XeF6) | Distorted Octahedral | 90 degrees (with distortions) |
As seen in the table, the Xenon Difluoride Bond Angle of 180 degrees is singular among these compounds. The square planar geometry of XeF4 and the twist octahedral geometry of XeF6 result from different hybridization states of the xenon atom and the number of fluorine atoms bond to it.
Applications and Reactivity of Xenon Difluoride
Xenon difluoride has several applications in chemistry, particularly in the field of fluorination reactions. Its reactivity is shape by its linear construction and the presence of lone pairs on the xenon atom. XeF2 can act as a fluorinating agent, transplant fluorine atoms to other molecules. This property makes it utile in the synthesis of respective organic and inorganic compounds.
Some of the key applications of XeF2 include:
- Fluorination of organic compounds to inclose fluorine atoms into molecules.
- Use in the synthesis of fluorinated polymers and materials.
- Application in the preparation of fluorinated pharmaceuticals and agrochemicals.
However, handle XeF2 requires precaution due to its high reactivity and potential to induce explosions when in contact with certain substances. Proper safety measures must be guide when work with this compound.
Note: Xenon difluoride is extremely reactive and should be care with care in a controlled environment. Always postdate safety protocols when working with this compound.
Conclusion
Xenon difluoride is a noteworthy compound that challenges traditional chemic theories. Its Xenon Difluoride Bond Angle of 180 degrees is a key factor in understanding its molecular geometry and reactivity. The linear structure of XeF2, resulting from sp3d hybridizing, contributes to its unique properties and applications in fluorination reactions. Comparing XeF2 with other xenon compounds highlights the variety of molecular geometries and bond angles in xenon chemistry. Understanding the bond and construction of XeF2 provides valuable insights into the chemistry of noble gases and their compounds, open up new avenues for research and applications.
Related Terms:
- fxe f bond angle
- xef2 bond pairs
- xef2 electron shape
- xef2 lewis polarity
- xef2 sign
- xef2 lewis bond